Dyestuff industry experts
ITMA AISA 2024
- Home
- About
- Online Q & A
- Products
- Applications
- Solution
- Service
- Our Brand
- Info
- Contact
Views: 49 Author: Site Editor Publish Time: 2025-07-11 Origin: Site
Acetic acid (chemical formula CH3COOH), commonly known as acetic acid, is an organic monobasic acid. Acetic acid with a content of 98% freezes at low temperatures and is commonly known as glacial acetic acid. The pH value of an aqueous solution of 1 mol/L acetic acid (equivalent to the concentration of vinegar) is 2.4.
Acetic acid should be the most commonly used acid in printing and dyeing processing: acid neutralization of alkaline pretreatment, pH adjustment of disperse dyes, acid dyes, cationic dyes, etc., pH adjustment of acid cellulase polishing, etc.
1. Acid can cause the strength of cotton fiber to decrease. Cotton cellulose will hydrolyze when it encounters acid. The principle is: the glucose residues of cellulose macromolecules are connected by 1,4-glycosidic bonds, and the glycosidic bonds are very sensitive to acid. Cotton cellulose is hydrolyzed when it encounters acid. When all the glycosidic bonds in the cellulose macromolecule are broken, glucose is generated. After cellulose is hydrolyzed, hydrolyzed cellulose is generated, the degree of polymerization decreases, and the strength also decreases. Therefore, acid can cause the strength of cotton fiber to decrease.
2. The influence of different acids on the strength of cotton fibers:
As shown in the figure above, hydrochloric acid and nitric acid have the greatest impact on the strength reduction of cotton fibers, followed by sulfuric acid, then sodium bisulfate and phosphoric acid. Formic acid has a much higher strength reduction than acetic acid, but many dyeing factories are still using formic acid for acid neutralization, probably because the cost of formic acid is lower than that of acetic acid. Taking all factors into consideration, acetic acid is the most suitable acid for cotton fiber processing.
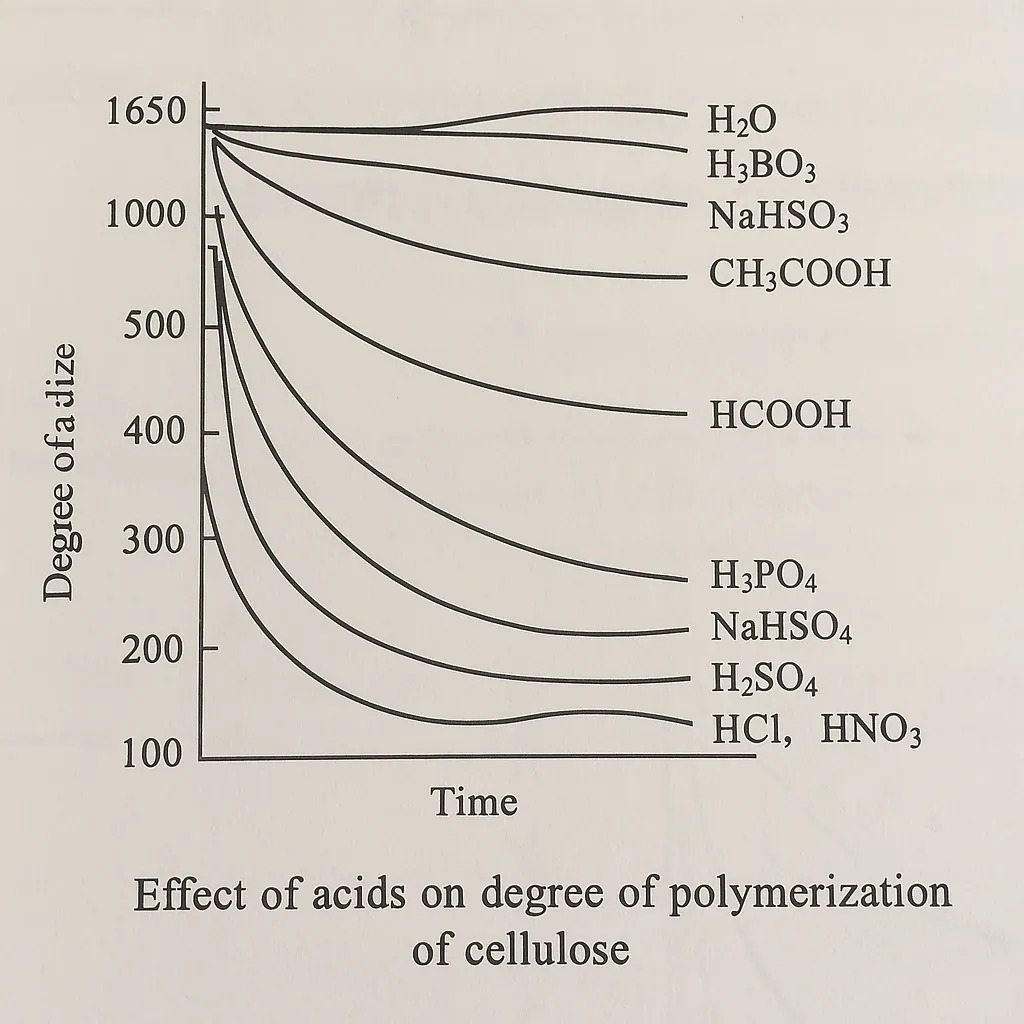
As shown in the figure above, hydrochloric acid and nitric acid have the greatest impact on the strength reduction of cotton fibers, followed by sulfuric acid, then sodium bisulfate and phosphoric acid. Formic acid has a much higher strength reduction than acetic acid, but many dyeing factories are still using formic acid for acid neutralization, probably because the cost of formic acid is lower than that of acetic acid. Taking all factors into consideration, acetic acid is the most suitable acid for cotton fiber processing
buffer solution: A mixed solution composed of weak acid and its salt, weak base and its salt, which can offset and reduce the influence of external strong acid or strong base on the pH value of the solution to a certain extent, thereby keeping the pH value of the solution relatively stable.
When dyeing disperse dyes, acid dyes, cationic dyes and other dyes, the pH is generally acidic. In order to achieve stable pH control, some factories will use acetic acid-sodium acetate buffer system to accurately control the pH to improve the reproducibility and success rate of dyeing.
The following is the preparation of acetic acid-sodium acetate buffer solution with commonly used pH:
Acetic acid-sodium acetate buffer (pH3.6)
Take 5.1g of sodium acetate, add 20ml of glacial acetic acid, and then dilute with water to 250ml.
Acetic acid-sodium acetate buffer (pH3.7)
Take 20g of anhydrous sodium acetate, add 300ml of water to dissolve, add 1ml of bromophenol blue indicator and 60-80ml of glacial acetic acid until the solution changes from blue to pure green, then dilute to 1000ml with water.
Acetic acid-sodium acetate buffer (pH3.8)
Take 13ml of 2mol/L sodium acetate solution and 87ml of 2mol/L acetic acid solution, add 0.5ml of copper sulfate solution containing 1mg of copper per 1ml, then dilute to 1000ml with water.
Acetic acid-sodium acetate buffer (pH4.5)
Take 18g of sodium acetate, add 9.8ml of glacial acetic acid, then dilute to 1000ml with water.
Acetic acid-sodium acetate buffer (pH4.6)
Take 5.4g of sodium acetate, add 50ml of water to dissolve, adjust the pH value to 4.6 with glacial acetic acid, and then dilute with water to 100ml.
Acetic acid-sodium acetate buffer (pH6.0)
Take 54.6g of sodium acetate, add 20ml of 1mol/L acetic acid solution to dissolve, and then dilute with water to 500ml.
Cotton fabric polishing is now widely used. Cellulase can remove the hairiness and tiny pilling on the surface of cotton fibers. The advantages of acetic acid in polishing are: because acetic acid will generate sodium acetate when it encounters alkaline substances, the surface of cotton fibers reaches the required pH value during the pH adjustment process, and because sodium acetate is generated when the alkali inside the fiber is neutralized, the pH value inside the fiber will be higher, so it helps to protect the fiber strength while achieving the effect of surface polishing.
The current pH safety standard for textiles generally requires 4-7.5, which is basically neutral and weakly acidic, and the test method is mostly extraction. When acetic acid is used for the last pH adjustment of textiles, because of the formation of acetic acid-sodium acetate buffer system, the alkali inside the fiber cannot be completely neutralized. The pH of the cloth surface tested with pH test paper is acidic, and the extraction method often shows that the pH is high. Therefore, the last pH is worth adjusting. Many factories choose to use citric acid or other organic acids with stronger acidity instead of acetic acid.
content is empty!